62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
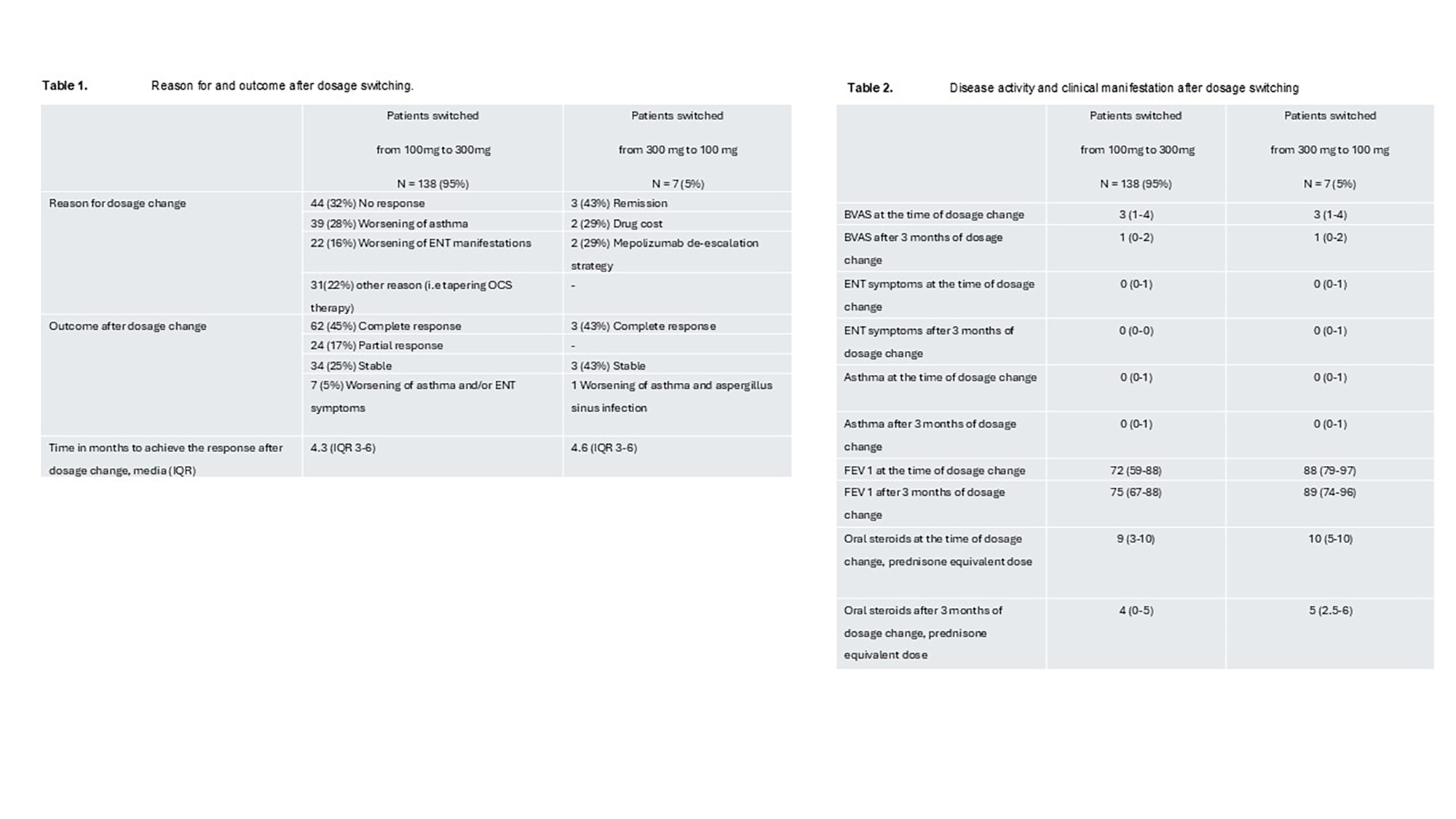
PO:35:218 | Switching the dose of mepolizumab in patients with Eosinophilic Granulomatosis with Polyangiitis (EGPA): a European retrospective multicentre real-world study
Edoardo Biancalana1, Irene Mattioli1, Emanuele Chiara2, Danilo Malandrino1, Maria Canfora1, Palmerini Miki1, Maria Paola Lisi1, Michela Gasparotto3, Paola Tomietto3, Maria Letizia Urban1, Giacomo Bagni1, Elena Silvestri1, Alessandra Bettiol3, Augusto Vaglio3, Giacomo Emmi2 | 1Department of Experimental and Clinical Medicine, University of Florence Firenze, Italy; 2Department of Medical, Surgery and Health Sciences, University of Trieste Trieste, Italy; 3Clinical Medicine and Rheumatology Unit, Cattinara University Hospital, Trieste, Trieste, Italy; 4Department of Biomedical, Experimental and Clinical Sciences Mario Serio, University of Florence Firenze, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
34
Views