62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
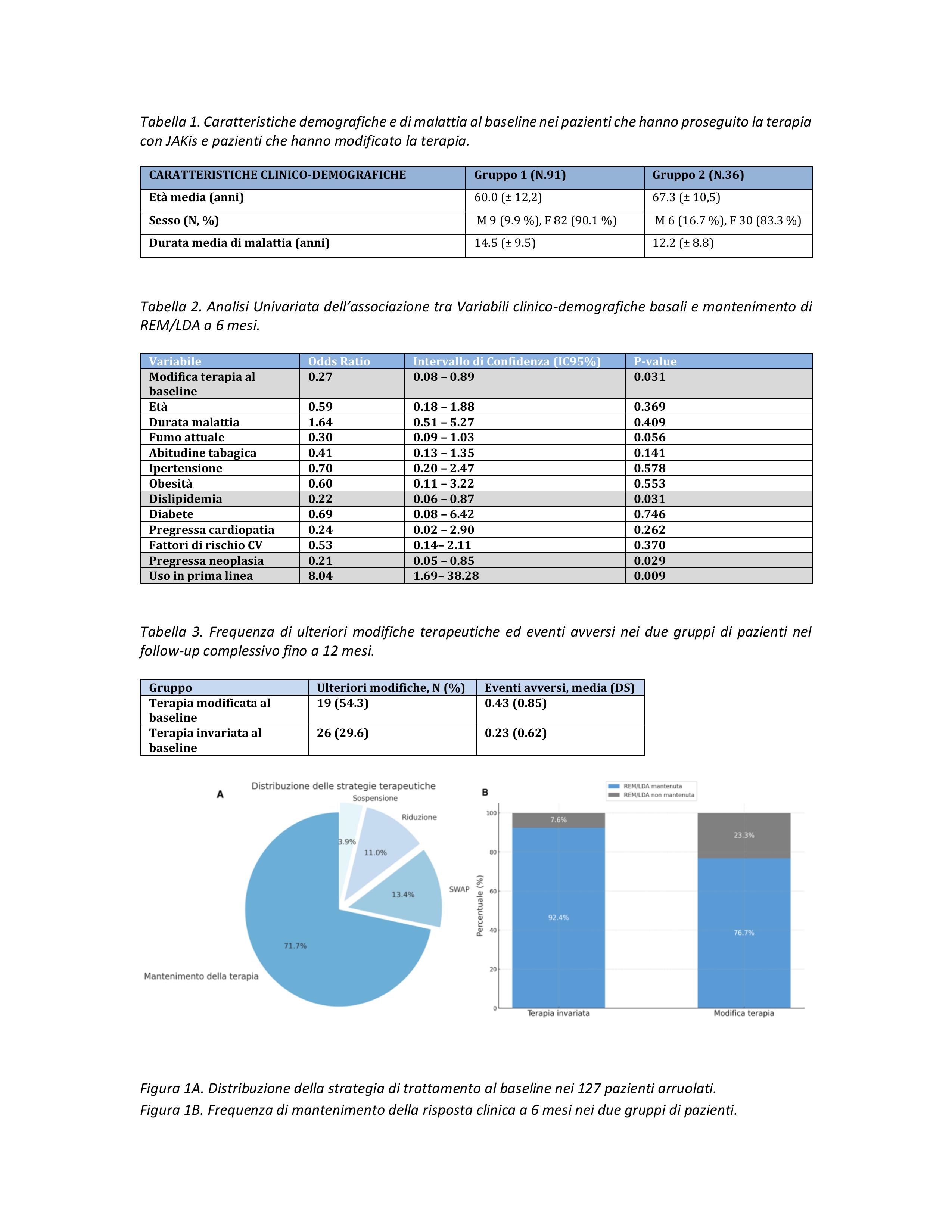
PO:07:095 | Clinical impact of therapeutic modification following changes in AIFA reimbursement criteria for JAK inhibitors in patients with RA in remission/low disease activity: analysis of the multicenter JAK-SWAP RER study in Emilia-Romagna
Marta Raschella1, Giulia Furia1, Martina Di Nunzio1, Francesco Luca Renzullo2, Francesco Girelli2, Federica Pignatti3, Francesco Ursini3, Licia Vultaggio4, Pierluigi Cataleta4, Elena Bravi5, Eugenio Arrigoni5, Francesca Bergossi6, Enrica Vandelli7, Massimo Reta6|7, Viola Magnani8, Andreina Manfredi8|9, Giuseppe Germanò8, Alessandra Bortoluzzi1, Marcello Govoni1, Ettore Silvagni1 | 1Unità Operativa Complessa di Reumatologia, Dipartimento di Scienze Mediche, Università degli Studi di Ferrara e Azienda Ferrara; 2UOS Reumatologia, Ospedale GB Morgagni, Forlì; 3UOC Reumatologia, Istituto ortopedico Rizzoli, Bologna; 4UO Reumatologia, AUSL Romagna Ospedale di Ravenna; 5UO Reumatologia, Azienda USL di Piacenza; 6Reumatologia Osp. Maggiore, Bologna; 7UO Medicina Interna ad Indirizzo Reumatologico Interaziendale SC AUSL Bologna, IRCCS Policlinico di SantOrsola, Bologna; 8U.O Reumatologia, Santa Maria Nuova di Reggio Emilia; 9Università degli studi di Modena e Reggio Emilia, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 25 November 2025
184
Views
Marta Raschella, Giulia Furia, Martina Di Nunzio, Francesco Luca Renzullo, Francesco Girelli, Federica Pignatti, Francesco Ursini, Licia Vultaggio, Pierluigi Cataleta, Elena Bravi, Eugenio Arrigoni, Francesca Bergossi, Enrica Vandelli, Massimo Reta, Viola Magnani, Andreina Manfredi, Giuseppe Germanò, Alessandra Bortoluzzi, Marcello Govoni, Ettore Silvagni