62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
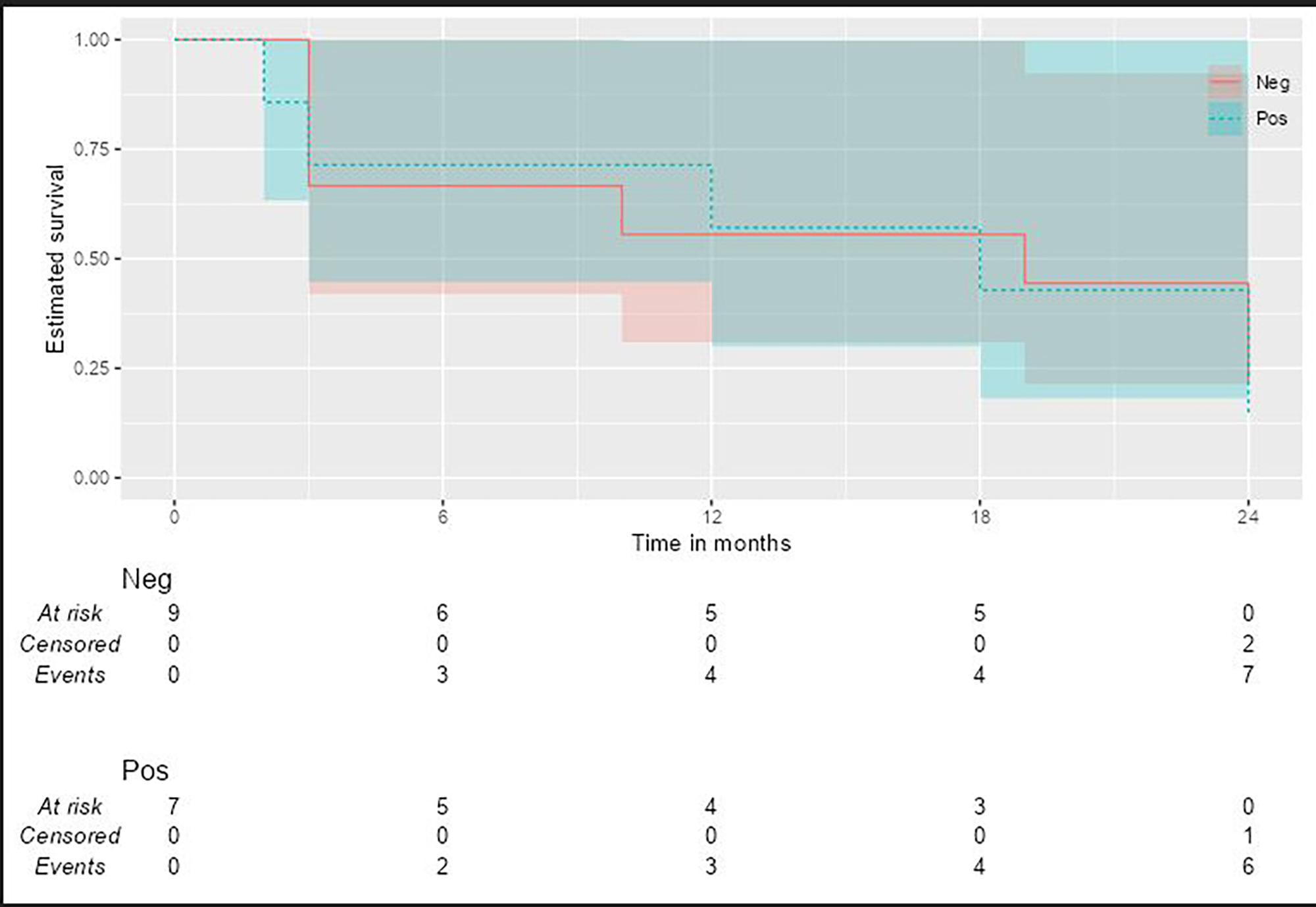
PO:35:234 | Sustained Vasculitis Quiescence and CS Reduction with Mepolizumab 100 mg
Francesca Mascia1, Angelo Fassio2, Marco Zurlo1, Davide Chesini3, Francesco Pollastri2, Giovanni Adami2, Davide Gatti4, Maurizio Rossini4, Matteo Maule5, Gianentrico Senna1, Marco Caminati1 | 1University of Verona - Allergology and Clinical Immunology Verona, Italy; 2Policlinico G. B. Rossi - Rheumatology Verona, Italy; 3University of Verona - Faculty of Medicine and Surgery Verona, Italy; 4University of Verona - Rheumatology Verona, Italy; 5Policlinico G. B. Rossi - Allergology and Clinical Immunology Verona, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
27
Views