62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
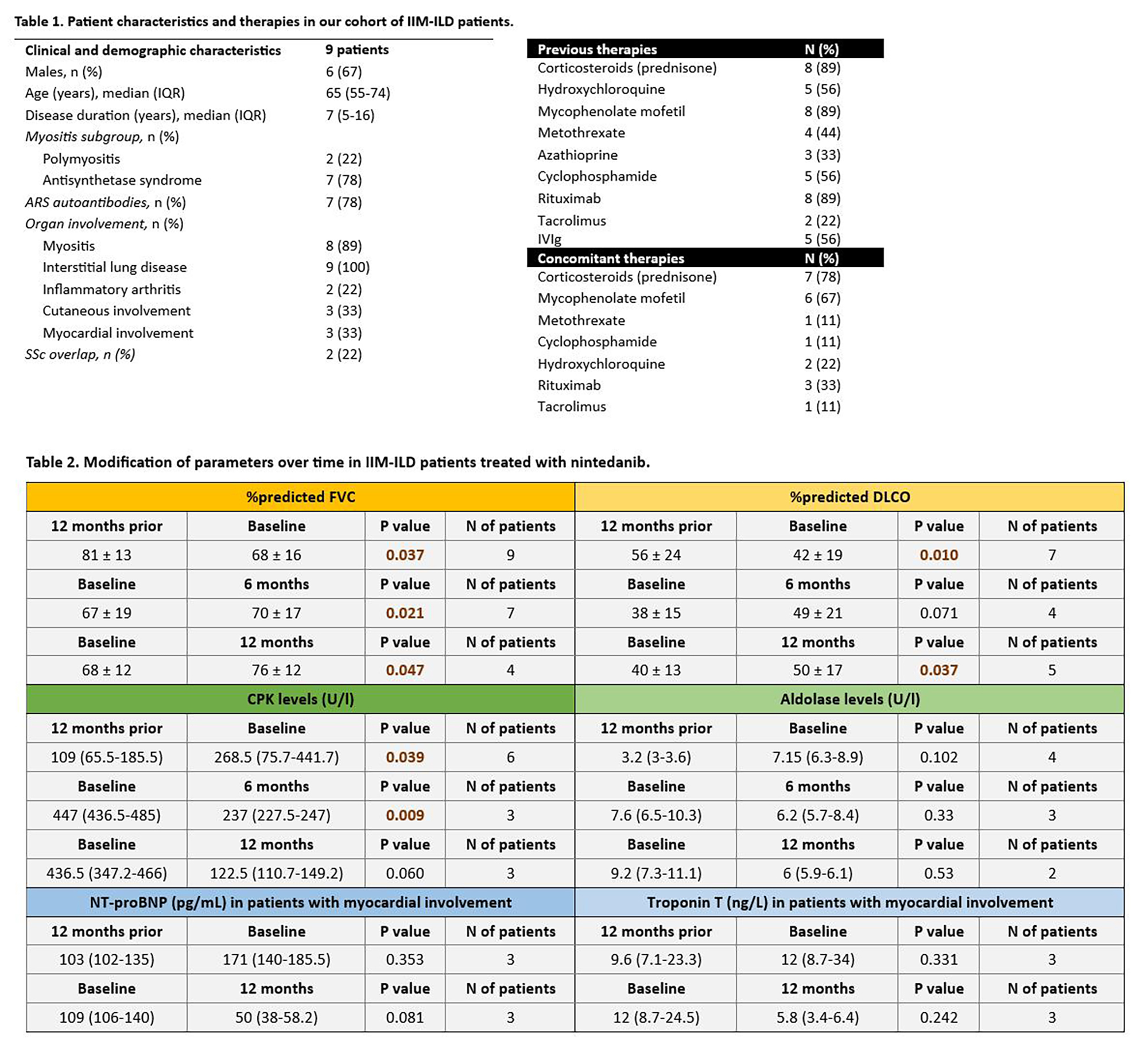
PO:24:063 | Interstitial lung disease in idiopathic inflammatory myopathy patients: efficacy and safety of nintedanib
Giulia Buonsante1|2|3, Giacomo De Luca1|2|3, Veronica Batani1|2, Lorenzo Dagna1|2|3, Marco Matucci-Cerinic1|2|3, Corrado Campochiaro1|2|3 | 1Unit of Immunology, Rheumatology, Allergy and Rare Diseases UNIRAR, IRCCS San Raffaele Hospital Milano, Italy; 2Inflammation, Fibrosis and aging Initiative INFLAGE, IRCCS San Raffaele Hospital Milano, Italy; 3Vita-Salute San Raffaele University, Milano, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
44
Views