62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
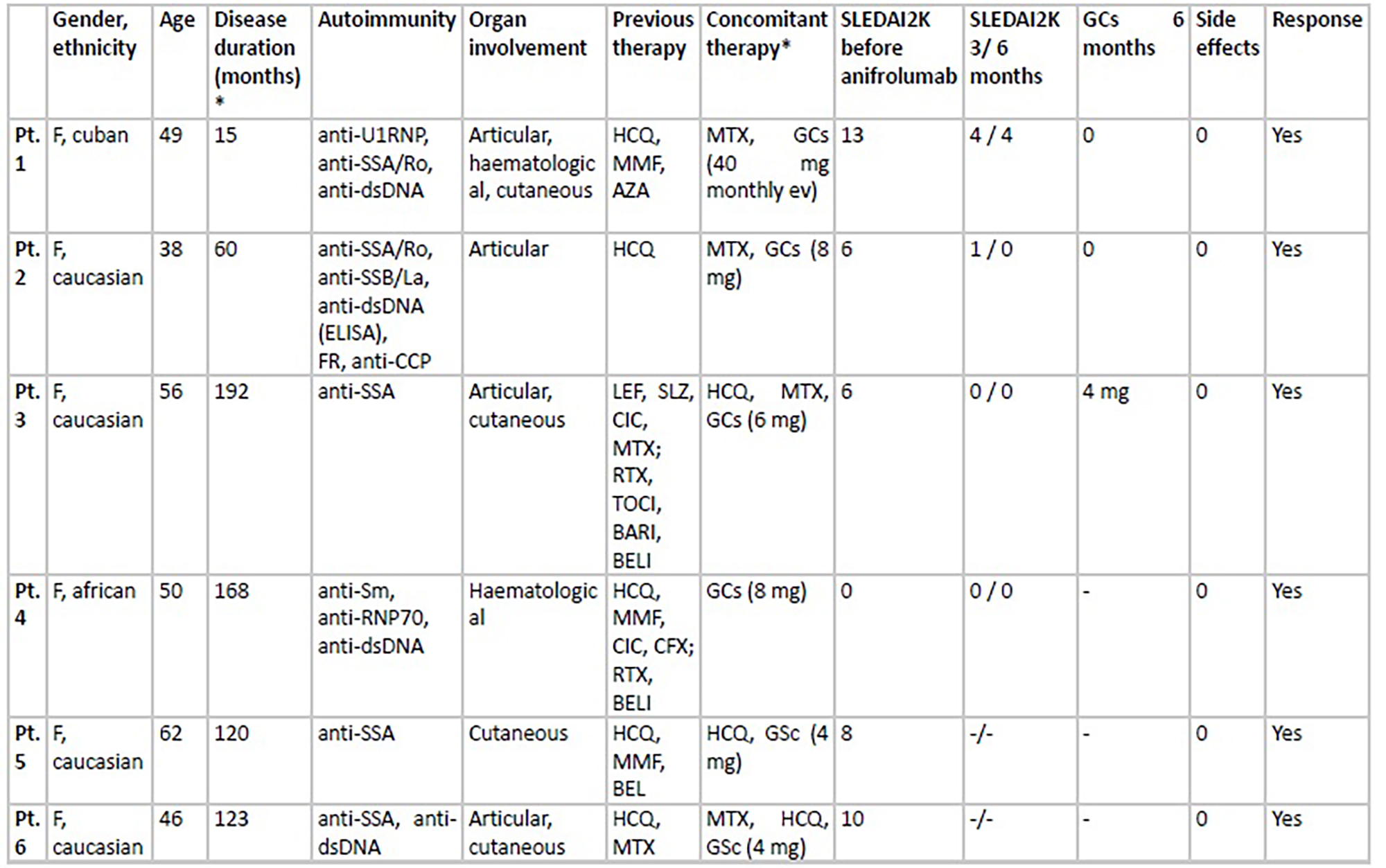
PO:18:264 | Anifrolumab in Systemic Lupus Erythematosus: Bridging Clinical Trial Data with Real-World Evidence
Giulia Cassone2, Filippo Santoro1, Caterina Vacchi2, Dilia Giuggioli2, Chiara Cabassi1 | 1UOC Reumatologia, AOU Policlinico di Modena, Università degli studi di Modena e Reggio Emilia Modena e Reggio Emilia, Italy; 2UOC Reumatologia, AOU Policlinico di Modena Modena, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
89
Views