62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
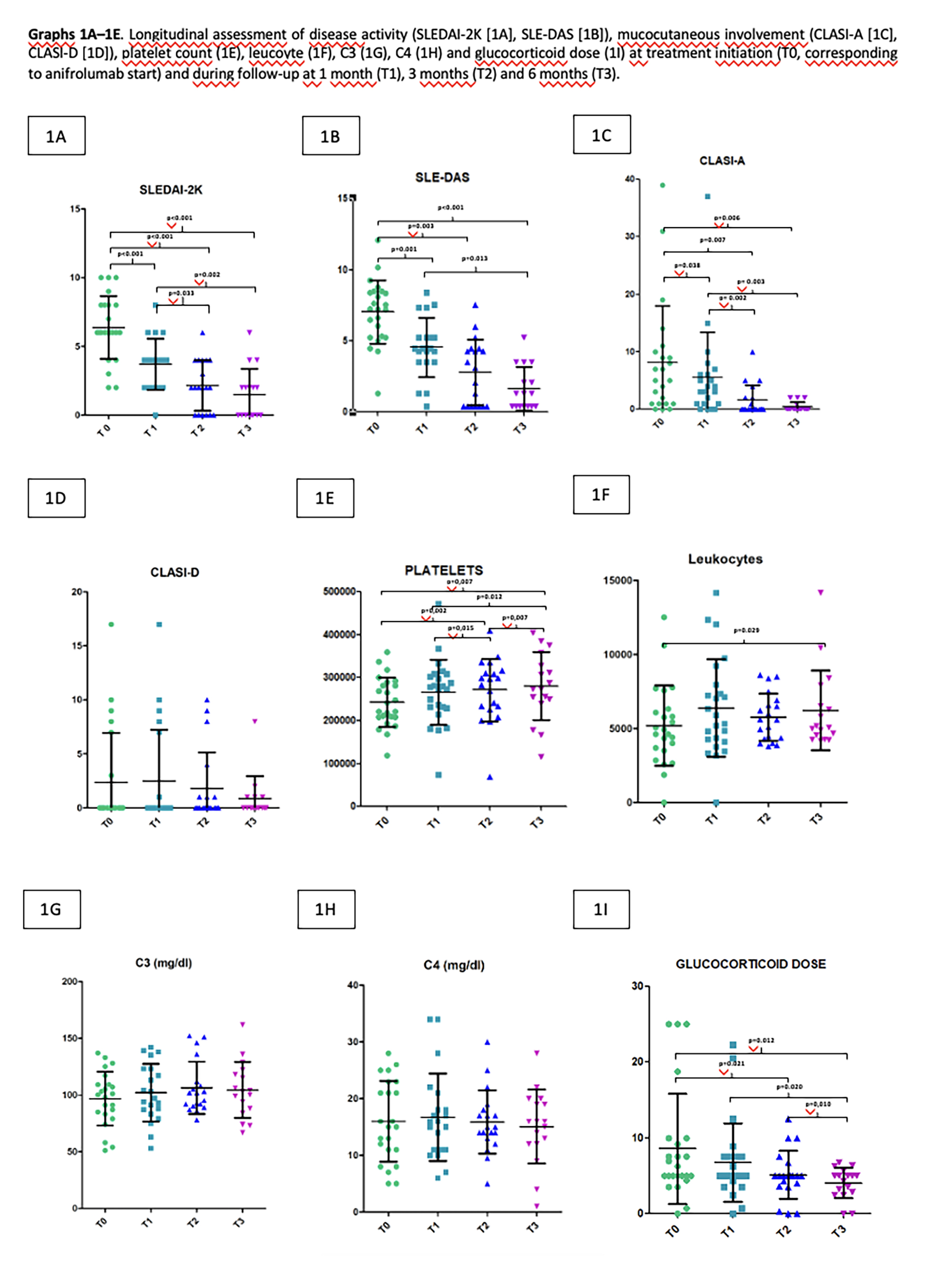
PO:16:227 | Anifrolumab for Systemic Lupus Erythematosus: Preliminary Safety and Efficacy Findings from the Italian Experience
Paola Bizioli1|2, Chiara Orlandi1|2, Claudia Barison1|2, Cesare Tomasi3, Alessia Caproli1, Silvia Piantoni1|2, Simona Signorini4, Emirena Garrafa4, Cecilia Nalli1, Micol Frassi1, Ilaria Cavazzana1|2, Micaela Fredi1|2, Franco Franceschini1|2 | 1Rheumatology and Clinical Immunology Unit - ERN ReCONNET, ASST Spedali Civili of Brescia, Italy; 2Department of Clinical and Experimental Sciences, University of Brescia, Italy; 3Department of Clinical and Experimental Sciences, University of Brescia, Italy; 4Laboratory of Clinical Chemistry, Department of Molecular and Translational Medicine, University of Brescia, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
50
Views