62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
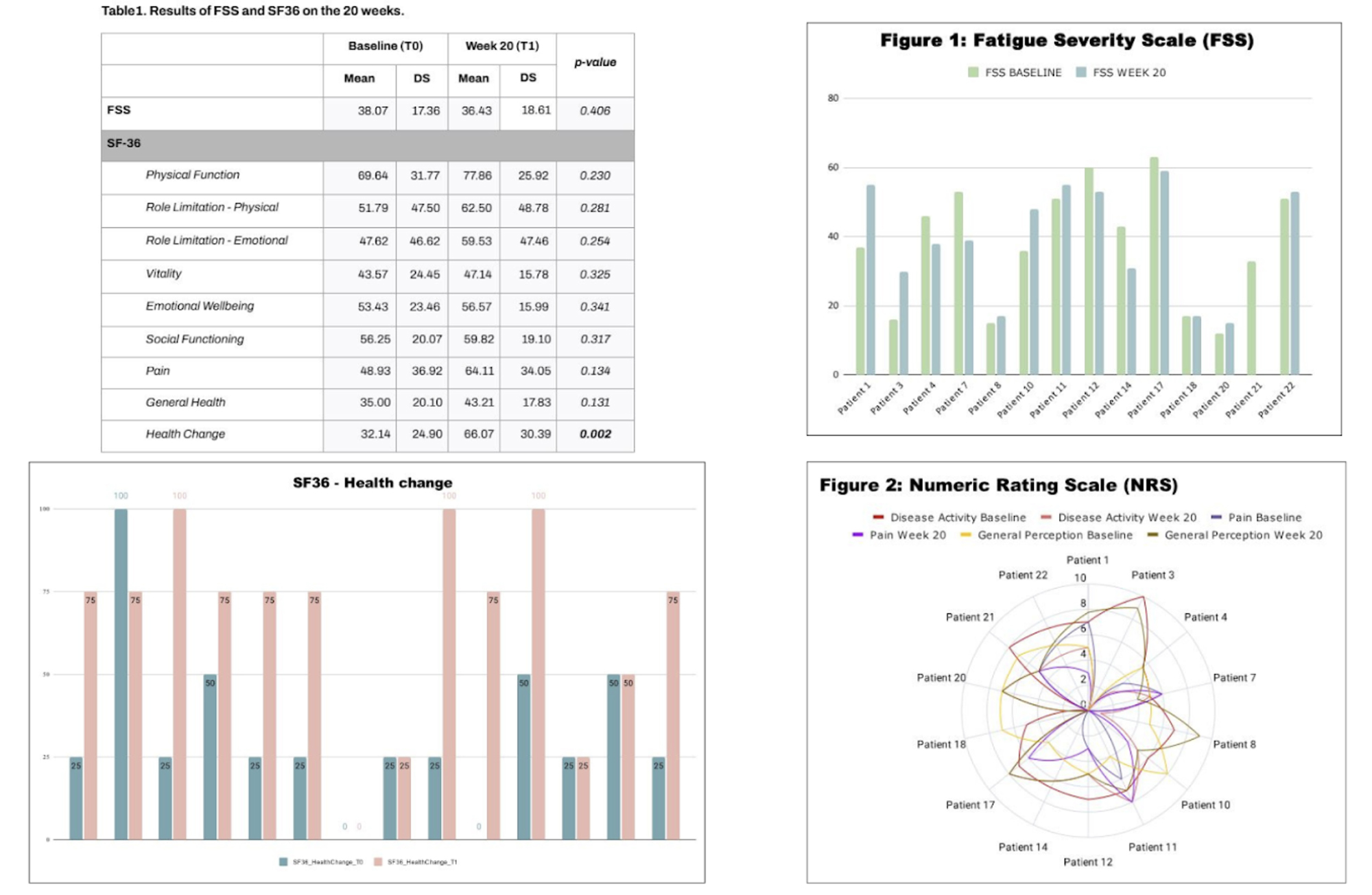
PO:16:225 | Belimumab in refractory and/or non-criteria manifestations of antiphospholipid syndrome: ad interim analysis on Quality-of-Life changes from the BLAST trial
Alessio Conti1, Lois Ira Capulong1, Irene Cecchi1, Elena Rubini1, Chiara Bova1, Massimo Radin1, Alice Barinotti1, Beatrice Ala1, Silvia Grazietta Foddai1, Daniela Rossi1, Roberta Fenoglio1, Savino Sciascia1 | 1Centro di Eccellenza Universitario per le Malattie Nefrologiche, Reumatologiche e Rare, S.G.Bosco -ASL Città di Torino, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
23
Views