62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
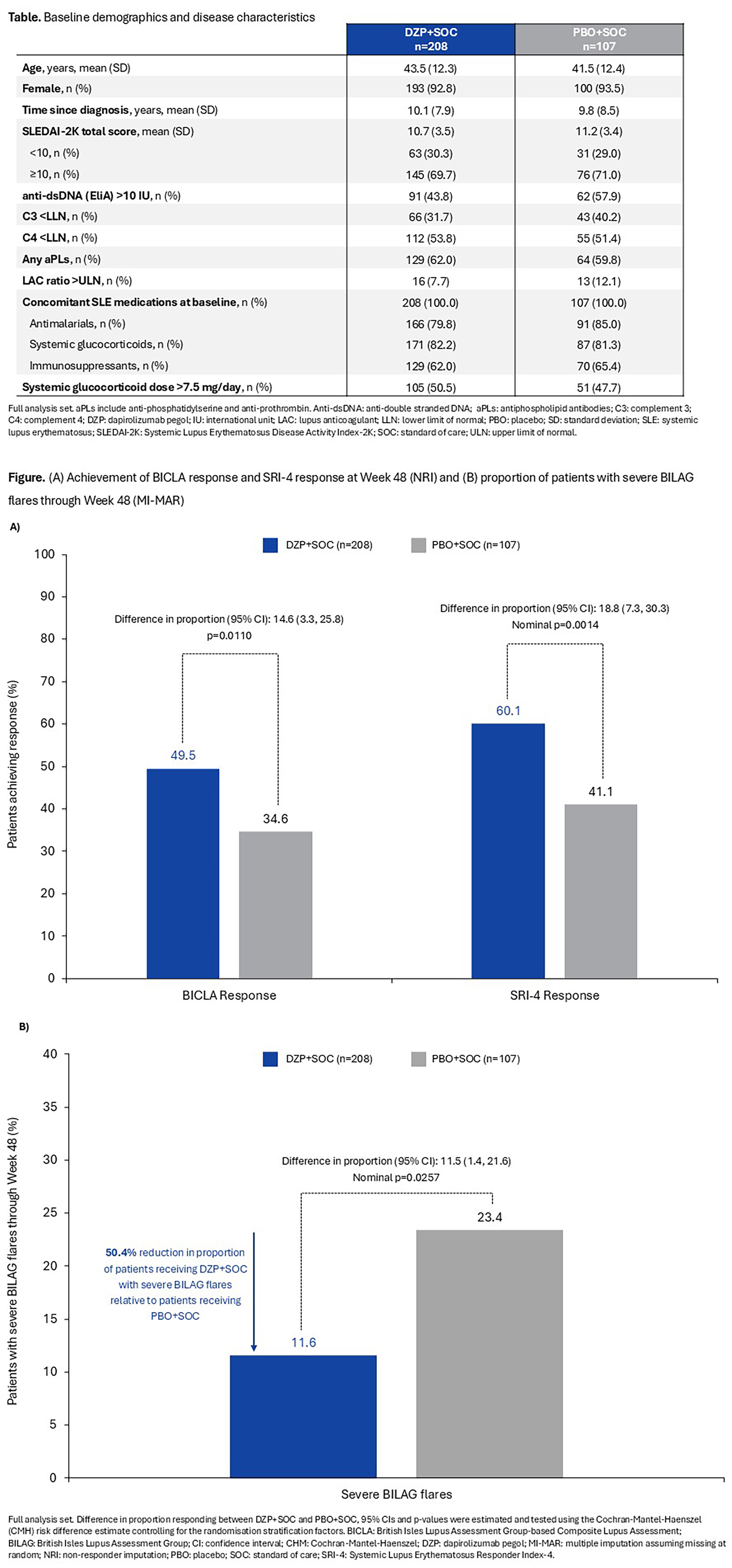
PO:14:193 | Dapirolizumab Pegol Demonstrated Significant Improvement in Systemic Lupus Erythematosus Disease Activity: Efficacy and Safety Results of a Phase 3 Trial
Megan EB Clowse1, Marta Mosca2, David A Isenberg3, Joan T Merrill4, Thomas Dörner5, Michelle Petri6, Edward M Vital7|8, Eric F Morand9, Teri Jimenez10, Stephen Brookes11, Janine Gaiha-Rohrbach12, Christophe Martin13, Annette Nelde14, Christian Stach15 | 1Duke University - Division of Rheumatology and Immunology Durham, USA; 2University of Pisa - Rheumatology Unit, Department of Clinical and Experimental Medicine Pisa, Italy; 3University College London - Department of Ageing, Rheumatology and Regenerative Medicine, Division of Medicine London, United Kingdom; 4Oklahoma Medical Research Foundation Oklahoma City, USA; 5Charité Universitätsmedizin Berlin - Department of Medicine/Rheumatology and Clinical Immunology Berlin Germany; 6Johns Hopkins University School of Medicine Baltimore, USA; 7University of Leeds - Leeds Institute of Rheumatic and Musculoskeletal Medicine Leeds, United Kingdom; 8Leeds Teaching Hospitals NHS Trust - NIHR Leeds Biomedical Research Centre Leeds, United Kingdom; 9Monash University - Centre for Inflammatory Diseases Melbourne, Australia; 10UCB Raleigh, USA; 11Biogen Maidenhead, United Kingdom; 12Biogen Cambridge, USA; 13UCB Slough, United Kingdom; 14Biogen Baar, Switzerland; 15UCB Monheim am Rhein, Germany
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
51
Views