62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
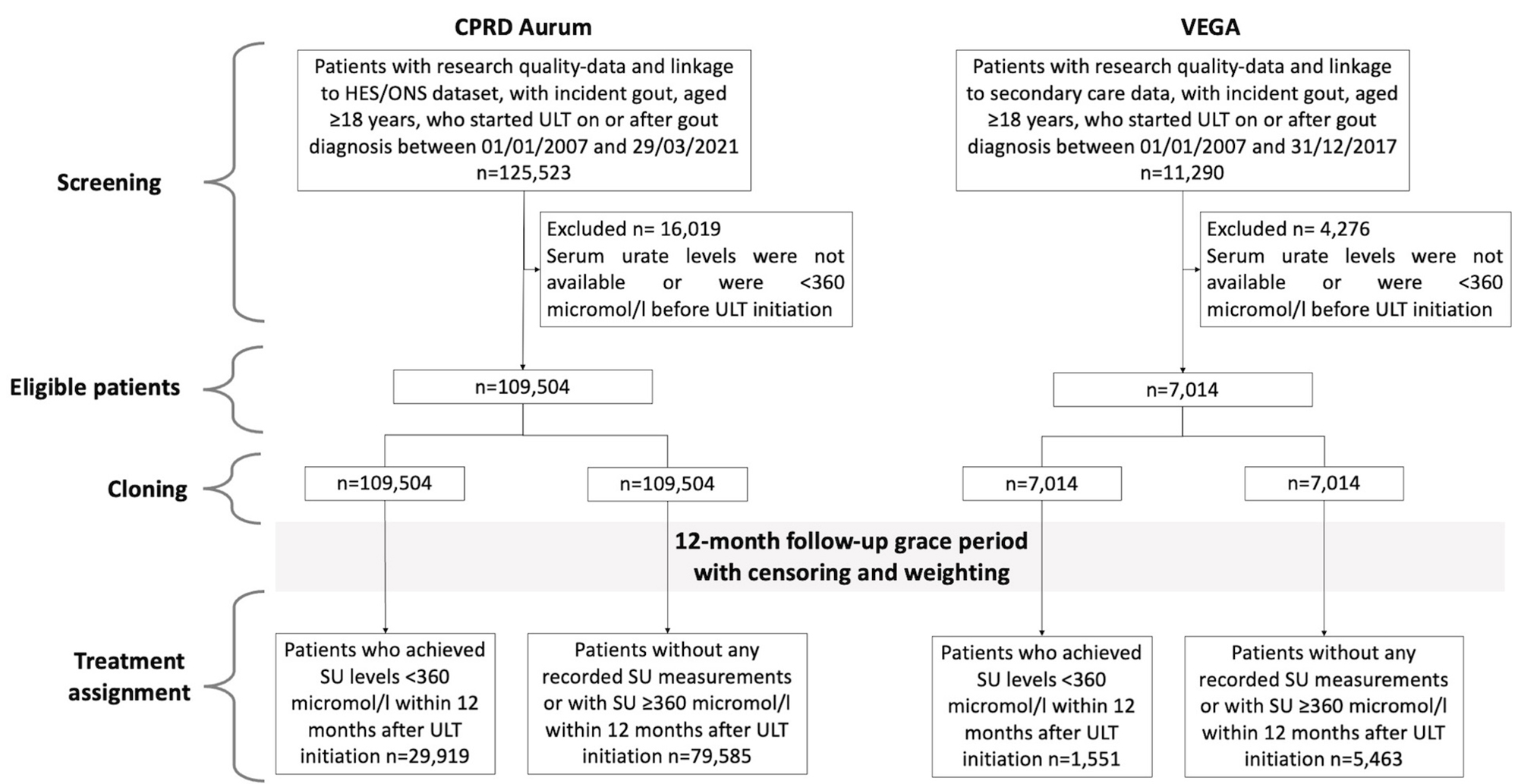
PO:12:179 | Cardiovascular outcomes of treat-to-target urate-lowering therapy in gout: emulated target trials
Edoardo Cipolletta1, Davide Rozza2, Abhishek Abhishek3 | 1Azienda Ospedaliero Universitaria delle Marche, Ancona, Italy; 2Società Italiana di Reumatologia, Milano, Italy; 3University of Nottingham Nottingham, United Kingdom
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
49
Views