62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
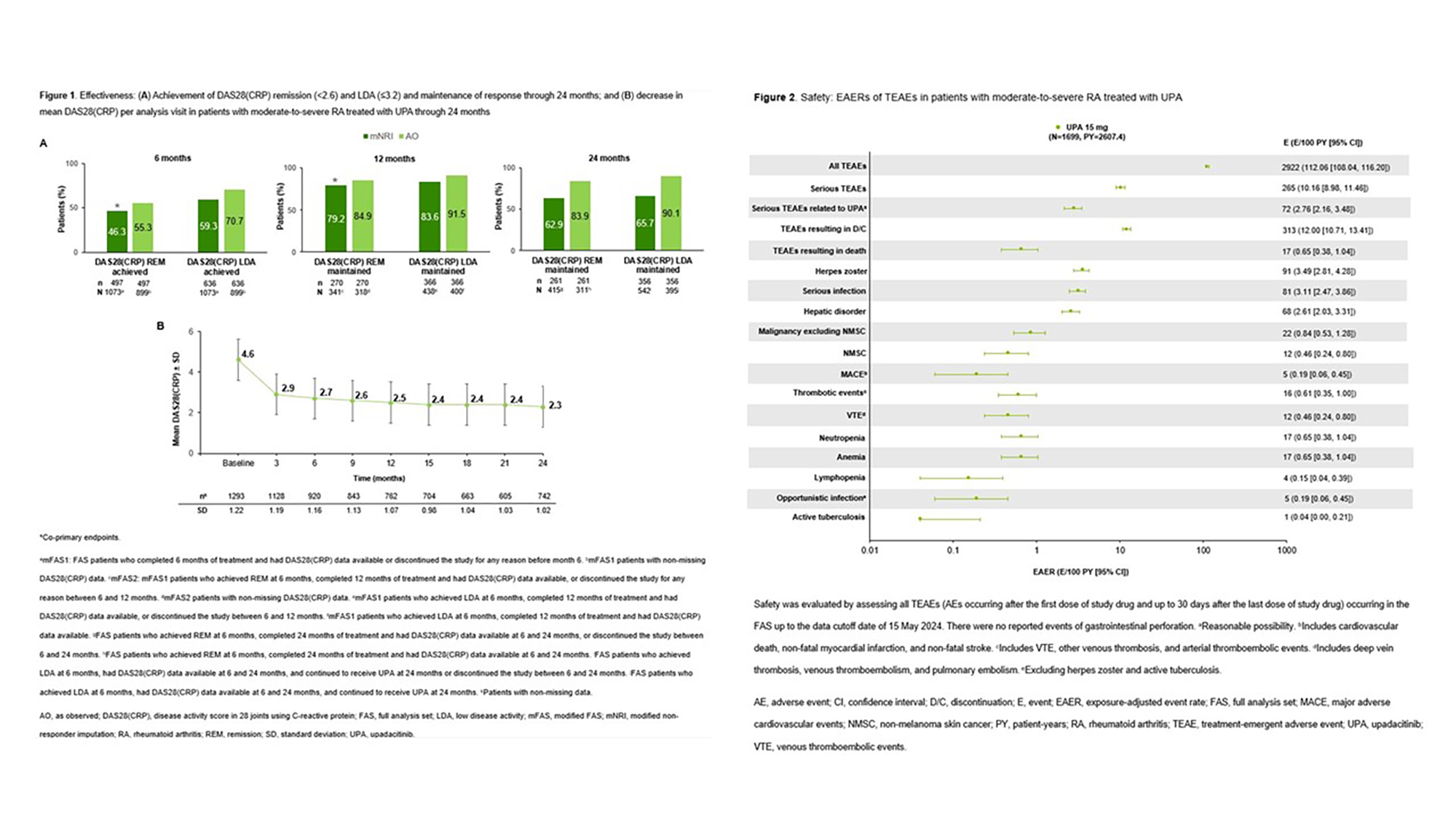
PO:07:108 | Achievement of Treatment Targets and Maintenance of Response with Upadacitinib in Patients with Moderate-to-Severe Rheumatoid Arthritis in a Real-world Setting: Final 2-year Outcomes from the UPHOLD Study
Enrico Marchetta1, Andrew Östör2|8, Eugen Feist3, Prodromos Sidiropoulos4, Jérôme Avouac5, Martin Rebella6|7, Rajaie Namas9, Erin Mcdearmon Blondell10, Ivan Lagunes10, Tianming Gao10, Tim Shaw10, Suzan Attar11 | 1AbbVie Srl Roma, Italy; 2Monash University and Emeritus Research, Melbourne, Victoria, Melbourne, Australia; 3Helios Department of Rheumatology and Clinical Immunology, Vogelsang-Gommern, Vogelsang Gommern, Germany; 4Faculty of Medicine, University of Crete, Heraklion, Heraklion, Greece; 5Service de Rhumatologie, Hôpital Cochin, AP HP Centre Université Paris Cité, Parigi, France; 6Departamento de Medicina, Facultad de Medicina, Universidad de la República, Montevideo, Montevideo, Uruguay; 7Unidad de Enfermedades Autoinmunes Sistémicas, MUCAM, Montevideo, Montevideo, Uruguay; 8Australian National University, Camberra, Australia; 9Medical Subspecialties Institute, Division of Rheumatology, Cleveland Clinic Abu Dhabi, United Arab Emirates; 10AbbVie Inc., North Chicago, Illinois, United States of America Chicago, USA; 11King Abdulaziz University, Jeddah, Saudi Arabia
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
37
Views