62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
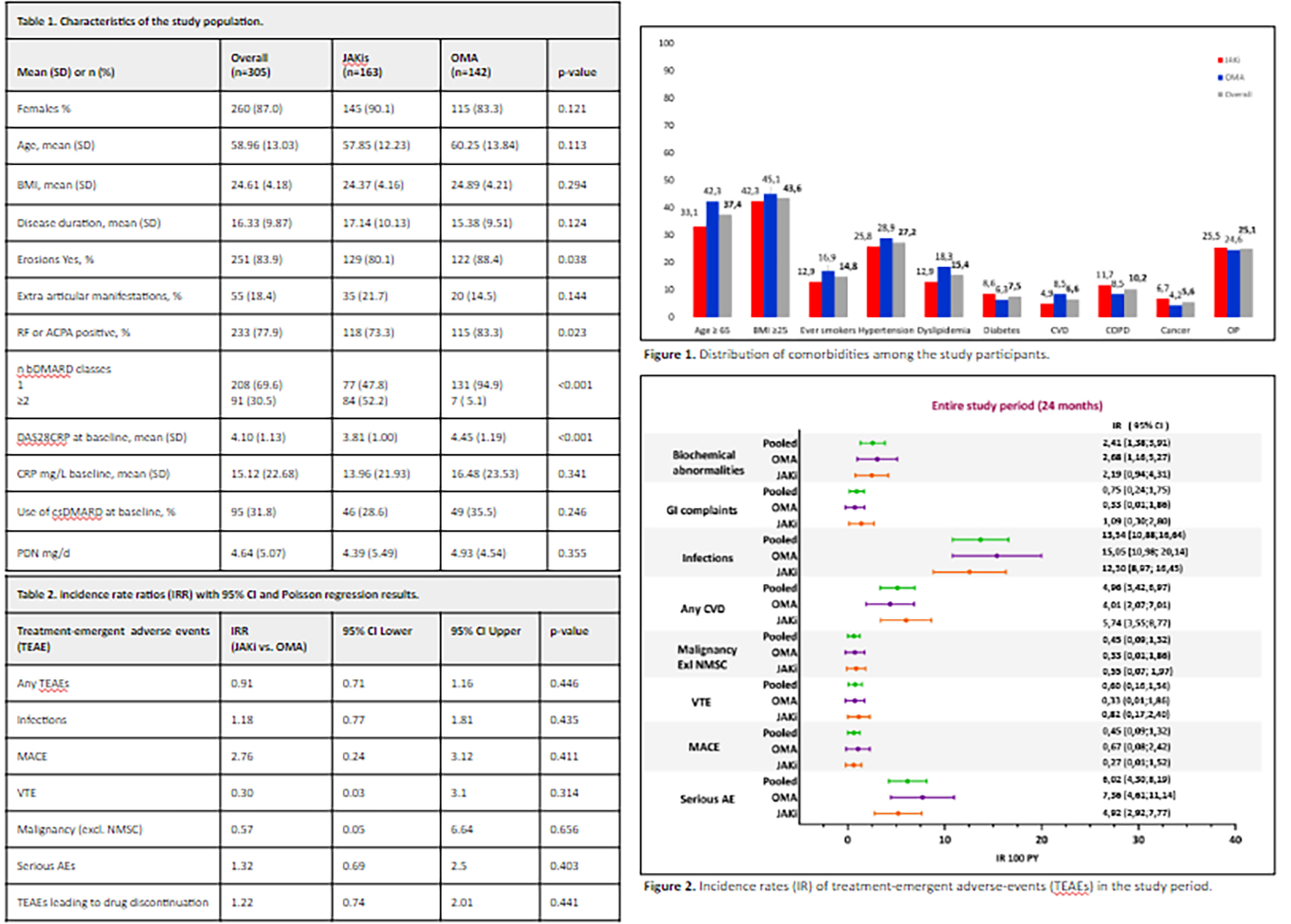
PO:06:081 | Safety of JAK inhibitors compared to non-TNF-targeted biologic therapies in rheumatoid arthritis patients who are inadequate responders to TNF inhibitors
Lorenzo Di Luozzo1, Alessandro Giollo1, Mariangela Salvato1, Francesca Frizzera1, Kiren Khalid1, Margherita Zen1, Andrea Doria1 | 1Azienda ospedaliera universitaria di Padova, unità di Reumatologia, Padova, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
44
Views