62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
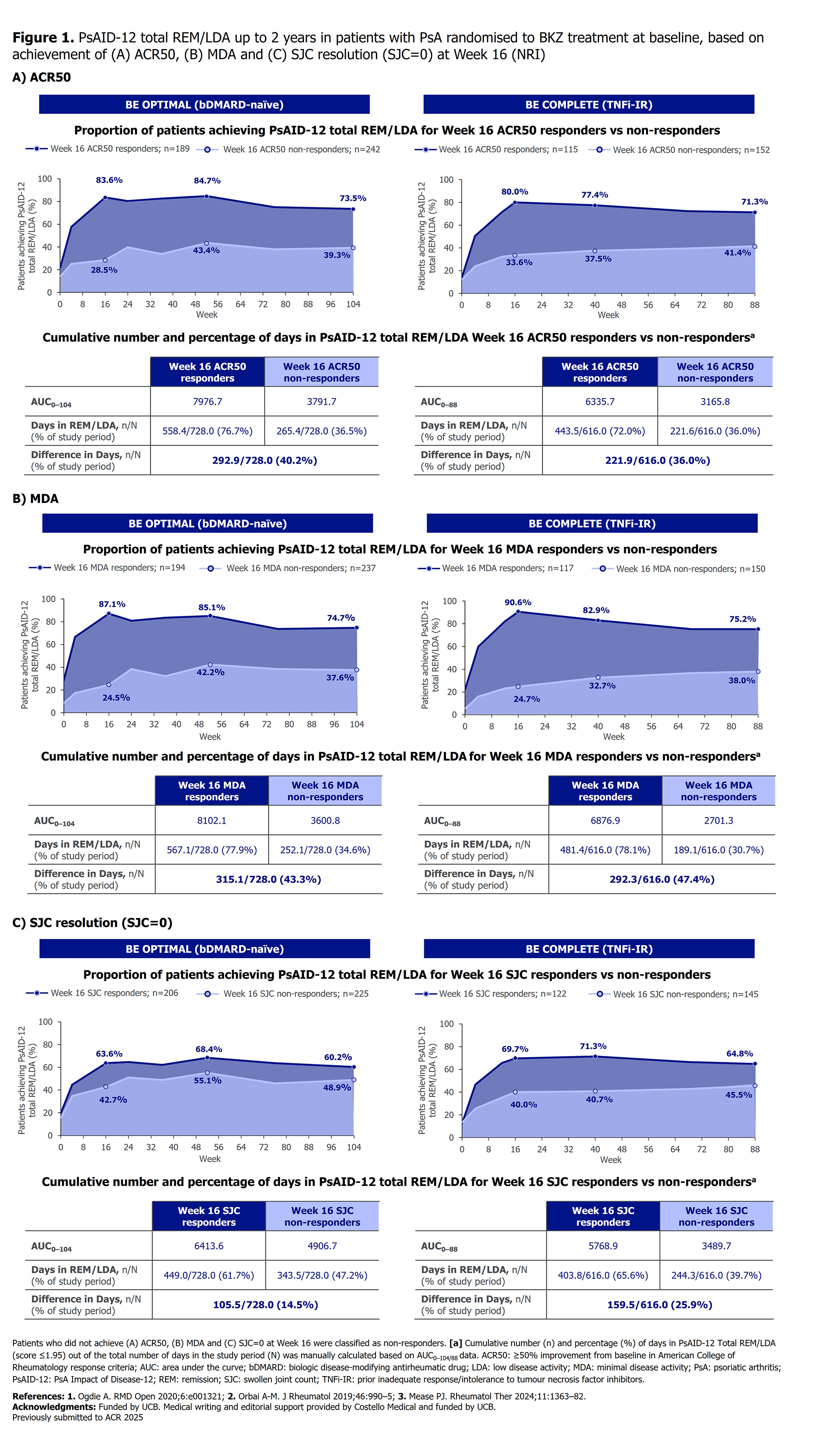
PO:04:046 | Achieving Early Clinical Response was Associated with Cumulative Benefits on Disease Impact up to 2 Years in Patients with Active Psoriatic Arthritis Treated with Bimekizumab
William Tillett1|2, Joseph F Merola3, Iain B Mcinnes4, Kenneth B Gordon5, Richard B Warren6, Patrick Healy7, Jérémy Lambert8, Heather Edens9, Barbara Ink10, Paola Volpe11, Laure Gossec12 | 1Royal National Hospital of Rheumatic Diseases Bath, United Kingdom; 2Department of Life Sciences, Centre for Therapeutic Innovation, University of Bath Bath, United Kingdom; 3Department of Dermatology and Department of Medicine, Division of Rheumatology, Utah Southwestern Medical Center Dallas, USA; 4College of Medical Veterinary and Life Sciences, University of Glasgow Glasgow, United Kingdom; 5Department of Dermatology, Medical College of Wisconsin Milwaukee, USA; 6Northern Care Alliance, NHS Foundation Trust and Manchester Academic Health Science Centre, University of Manchester Manchester, United Kingdom; 7UCB Morrisville, USA; 8UCB Colombes, France; 9UCB Smyrna, USA; 10UCB Slough, United Kingdom; 11UOC Reumatologia, P.O. Spirito Santo Pescara, Italy; 12Sorbonne Universite and Pitie-Salpetriere Hospital Paris, France
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
37
Views