62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
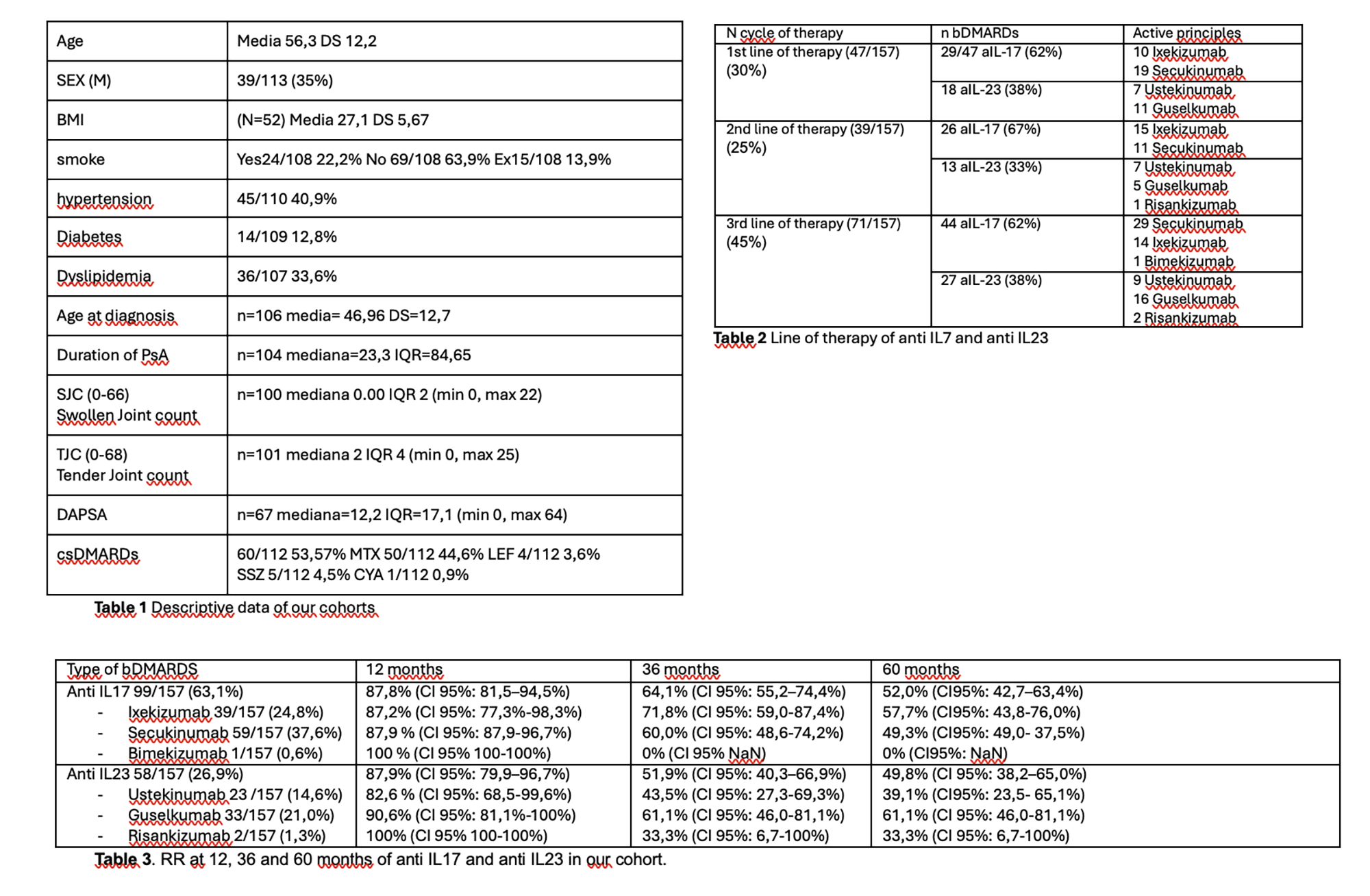
PO:02:030 | Retention Rate of Non-anti-TNF Therapies and primary failure in monocentric cohort of patients with Psoriatic Arthritis
Alba Chiara Pozzi1, Sofia Barezzani2, Andrea Galli2, Cesare Tomasi1, Micol Frassi1, Ilaria Cavazzana1, Franco Franceschini1 | 1Rheumatology and Clinical Immunology Unit ERN-ReCONNECT, ASST Spedali Civili, University of Brescia Brescia, Italy; 2Facoltà di Medicina e Chirurgia, University of Brescia Brescia, Italy
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
28
Views