62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
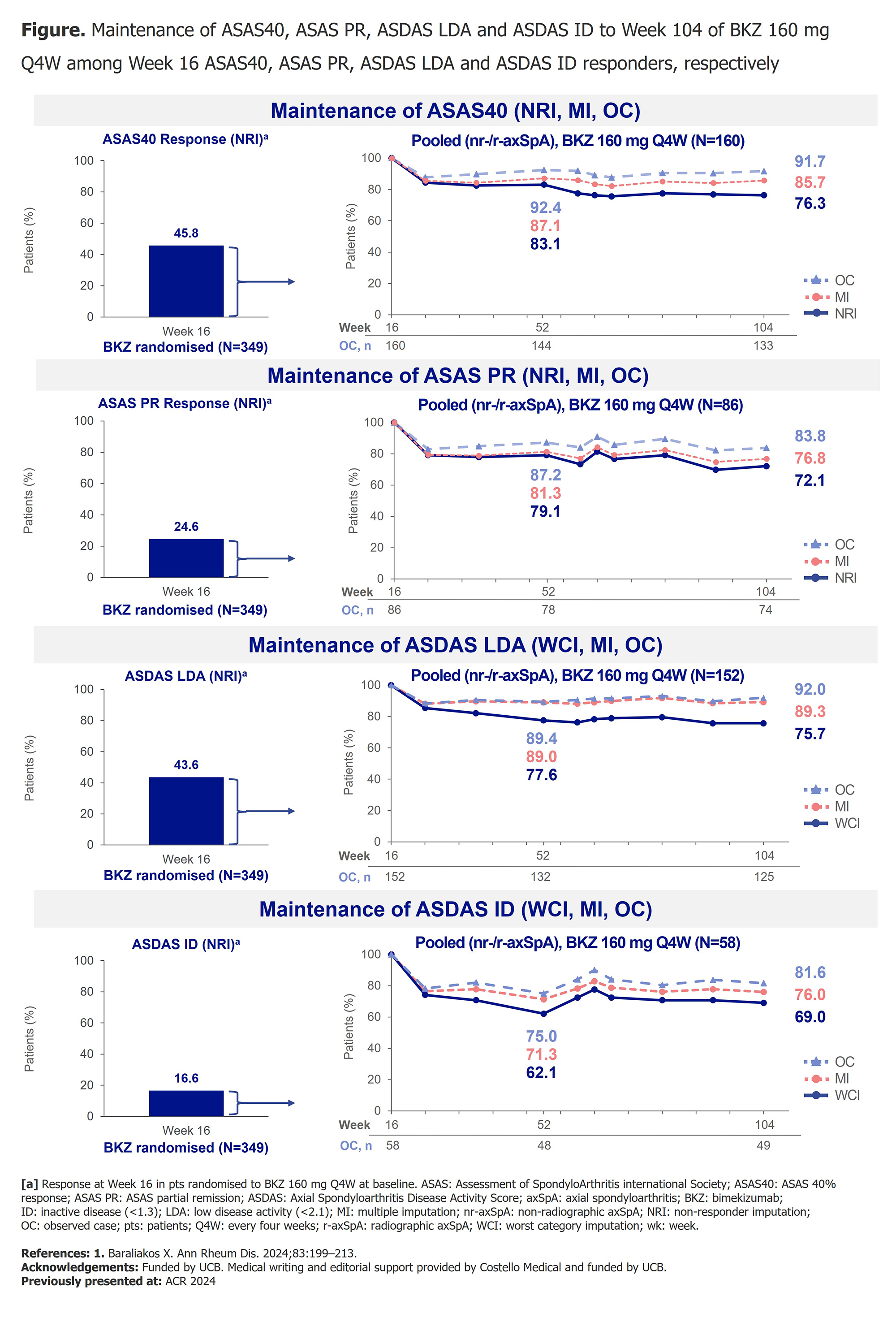
PO:02:022 | Bimekizumab Maintained Stringent Clinical Responses over 2 Years in Patients with Axial Spondyloarthritis: Results from Two Phase 3 Studies
Fabian Proft1, Désirée Van Der Heijde2, Sergio Schwartzman3|4|5, Joerg Ermann6, Alexander Marten7, Ute Massow7, George Stojan8, Vanessa Taieb9, Diana Voiniciuc10, Astrid Van Tubergen11, Victoria Navarro-Compán12, Simone Angioni13, Xenofon Baraliakos14 | 1Charité-Universitätsmedizin Berlin; Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Germany; 2Department of Rheumatology, Leiden University Medical Center Leiden, The Netherlands; 3Weill Cornell Medical Center New York, USA; 4New York Presbyterian Hospital New York, USA; 5Hospital for Special Surgery New York, USA; 6Division of Rheumatology, Inflammation and Immunity, Brigham and Women's Hospital and Harvard Medical School Boston, USA; 7UCB Monheim am Rhein Germany; 8UCB Atlanta, USA; 9UCB Colombes, France; 10UCB Slough, United Kingdom; 11Department of Medicine, Division of Rheumatology, Maastricht University Medical Center Maastricht, The Netherlands; 12Department of Rheumatology, La Paz University Hospital, IdiPaz Madrid, Spain; 13UCB Milan, Italy; 14Rheumazentrum Ruhrgebiet Herne, Ruhr-University Bochum, Germany
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
24
Views