62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
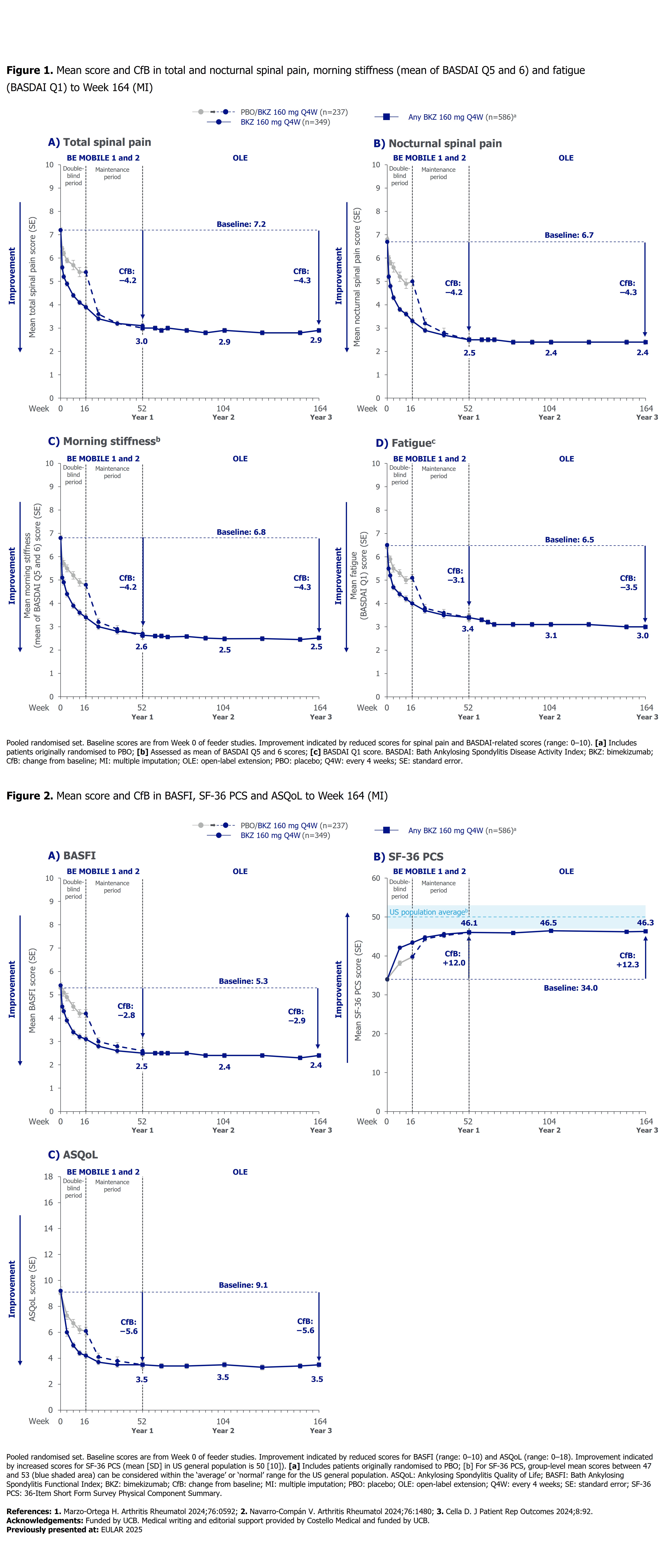
PO:02:021 | Sustained Improvements with Bimekizumab in Pain, Morning Stiffness, Fatigue, Physical Function and Health-Related Quality of Life in Patients with Axial Spondyloarthritis: 3-Year Results from Two Phase 3 Studies
Victoria Navarro-Compán1, Uta Kiltz2|3, Philip J Mease4, Maureen Dubreuil5, Karl Gaffney6, Atul Deodhar7, Christine De La Loge8, Diana Voiniciuc9, Jason Coarse10, Ilaria Spadafora11, Helena Marzo-Ortega12 | 1La Paz University Hospital, IdiPaz, Department of Rheumatology Madrid Spain; 2Ruhr-University Bochum Bochum Germany; 3Rheumazentrum Ruhrgebiet Herne, Germany; 4Department of Rheumatology, Swedish Medical Center and Providence St. Joseph Health and University of Washington Seattle, USA; 5Boston University School of Medicine, Department of Rheumatology Boston, USA; 6Norfolk and Norwich University Hospital NHS Trust, Department of Rheumatology Norfolk, United Kingdom; 7Oregon Health and Science University, Division of Arthritis and Rheumatic Diseases Portland, USA; 8UCB Brussels, Belgium; 9UCB Slough, United Kingdom; 10UCB Morrisville, USA; 11UCB Milan, Italy; 12NIHR Leeds Biomedical Research Centre, University of Leeds Leeds, United Kingdom
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
26
Views