62nd National Congress of the Italian Society of Rheumatology
Vol. 77 No. s1 (2025): Abstract book of the 62th Conference of the Italian Society for...
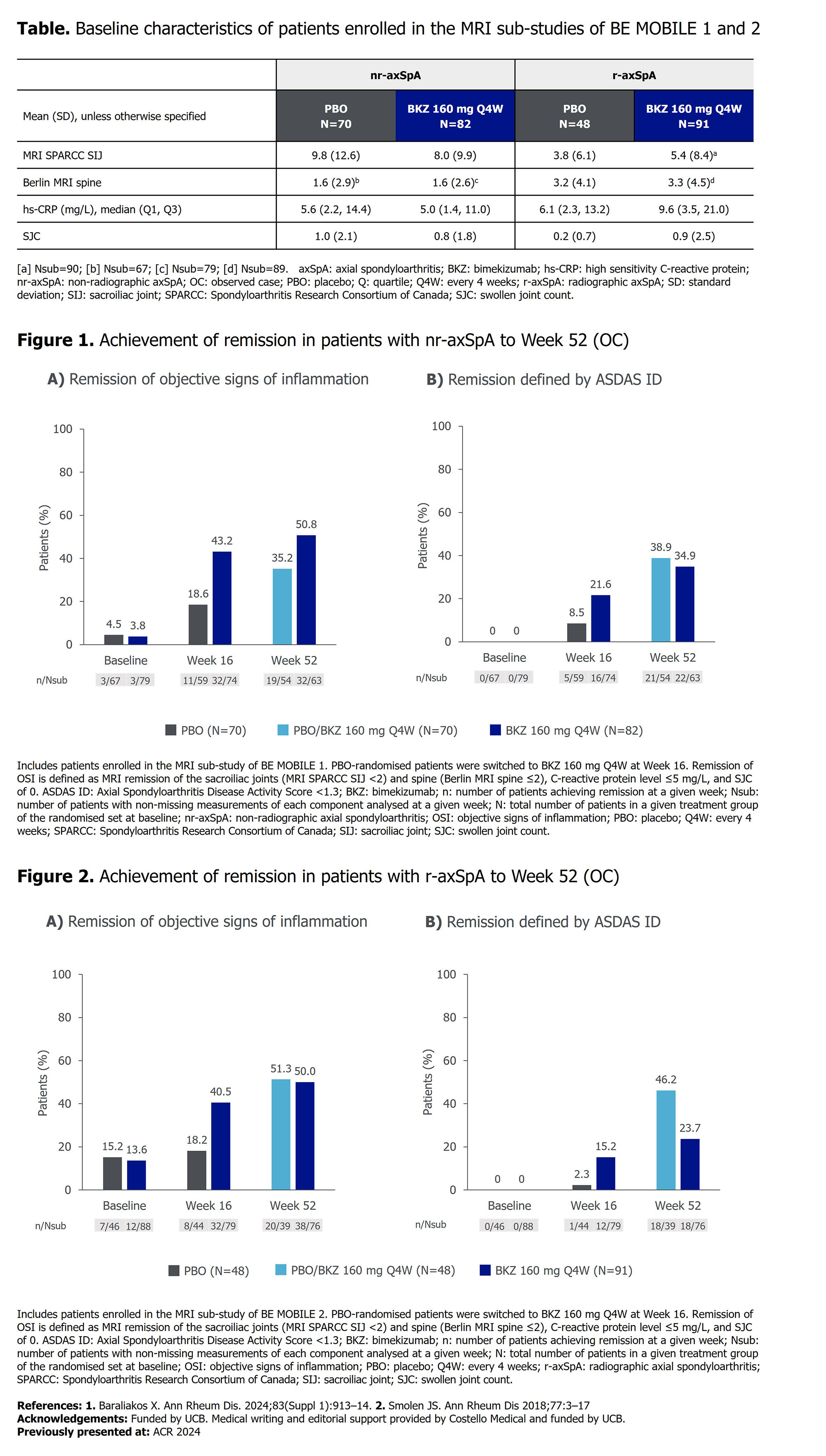
PO:01:011 | Achievement of Remission Defined by Absence of Objective Signs of Inflammation Versus ASDAS ID in Patients with Active Axial Spondyloarthritis Treated with Bimekizumab: 52-Week Results from Two Phase 3 Studies
Lianne S Gensler1, Helena Marzo-Ortega2, Vanessa Taieb3, Diana Voiniciuc4, Alexander Marten5, George Stojan6, Mindy Kim6, Paolo Marsico7, Martin Rudwaleit8 | 1Department of Medicine/Rheumatology, University of California San Francisco, USA; 2NIHR Leeds Biomedical Research Centre, University of Leeds Leeds, United Kingdom; 3UCB Colombes, France; 4UCB Slough, United Kingdom; 5UCB Monheim am Rhein Germany; 6UCB Atlanta, USA; 7UCB Milan, Italy; 8University of Bielefeld, Klinikum Bielefeld Bielefeld Germany
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 18 March 2026
27
Views