Brachio-cervical inflammatory myopathy associated with systemic sclerosis. Case series and review of literature
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Accepted: 7 April 2021
Authors
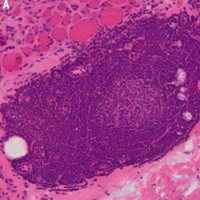
This study was aimed at describing a case series of brachio-cervical inflammatory myopathy (BCIM) associated with systemic sclerosis (SSc), due to its rarity and limited coverage in published data. Another aim was to provide a literature review. We reported four cases of BCIM-SSc from our tertiary center. In addition, we researched the literature and found six articles featuring 17 patients who fit this phenotype. We pooled all cases and reported their features. Most patients were female and had limited SSc, and the median time of BCIM presentation was three years after SSc diagnosis. Asymmetric muscle involvement, scapular winging, dropped head, axial weakness, camptocormia, dysphagia, and dermatomyositis stigmas were common features. All patients had esophageal involvement. Most had positive antinuclear antibody results, a scleroderma pattern in their capillaroscopy images, elevated serum creatine phosphokinase, myopathic electrophysiology, and muscle involvement in magnetic resonance imaging. Muscle histopathological findings varied widely, but in general all showed the presence of lymphoid infiltrates, muscle atrophy, increased MHC-I expression, MAC deposits, vasculopathy, and muscle fiber necrosis. The response to immunosuppressive therapy was highly irregular. BCIM-SSc is a rare disorder that shares many similar phenotypes among the described cases, but has a highly heterogeneous response to treatment. At present, more data on the physiopathology, clinical features, and treatment is still needed.











